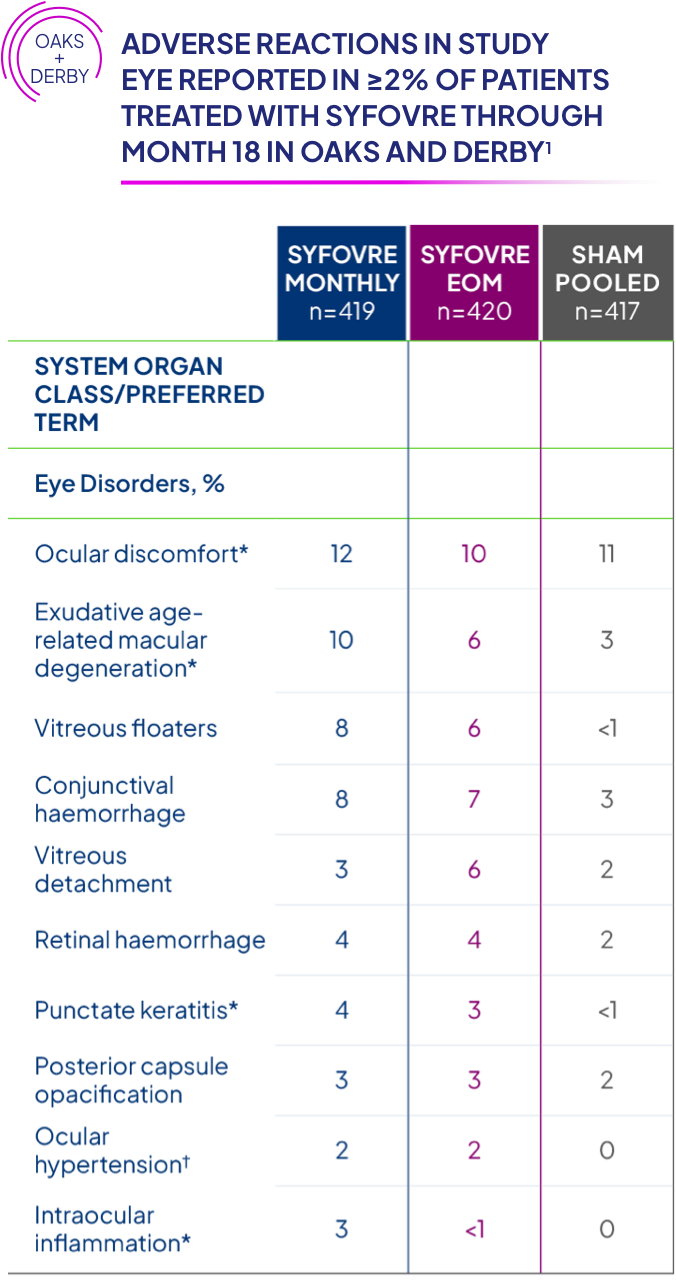
Who should NOT receive SYFOVRE® (pegcetacoplan injection)?
Do not receive SYFOVRE if you have an infection or active swelling in or around your eye that may include pain and redness, or are allergic to pegcetacoplan or any ingredients in SYFOVRE. SYFOVRE can cause serious allergic reactions such as trouble breathing, tongue, face, lips, or mouth swelling, rashes, and hives.
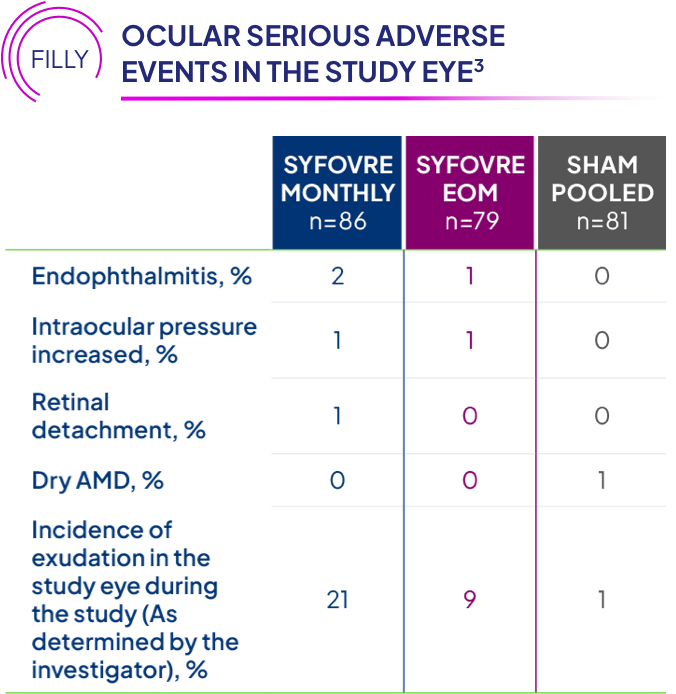
Who should NOT receive SYFOVRE® (pegcetacoplan injection)?
Do not receive SYFOVRE if you have an infection or active swelling in or around your eye that may include pain and redness, or are allergic to pegcetacoplan or any ingredients in SYFOVRE. SYFOVRE can cause serious allergic reactions such as trouble breathing, tongue, face, lips, or mouth swelling, rashes, and hives.












 Back to top
Back to top